Determining the true risk of COVID-19 for young people
3rd Sept, 2021
Sydney’s latest COVID-19 outbreak has seen a number of young people hospitalised after testing positive to the virus, with some younger than 20 in ICU [1]. At first glance, this appears to be an alarming development. But we need to know the full story behind these young people's health status before we can properly assess the risk to young people generally from the virus.
The government and the media are not always giving us that information.
The tragic story of the 15-year-old boy whose death was linked to COVID-19 in mid-August is a case in point. Many news outlets initially reported that he died from COVID-19, but it soon became clear that he had been admitted with a severe underlying illness [2]. And yet this incident is still shown on the COVID deaths list.
The Informed Medical Options Party calls on the NSW government to provide details of the underlying health status not only of adolescents in ICU but ALL patients in ICU with COVID, and to publish these details on a regular basis. Additionally, we ask for publication of the status of those that the health department has “hospitalised for isolation purposes and not severe disease” [3].
This is particularly important given the government’s push to vaccinate young adults, most of whom face greater risk from vaccine injury than the virus. The latest risk assessment from the Australian government shows that even in a high exposure risk setting, people aged 18-29 were more likely to get a blood clot from the AstraZeneca vaccine (1.9 clots for every 100,000 vaccines) than die from COVID-19, the chance of which is only 0.1 per 100,000 [4].
Back in June this year, the US FDA published, “revisions to the patient and provider fact sheets for the Moderna and Pfizer-BioNTech COVID-19 vaccines regarding the suggested increased risks of myocarditis (inflammation of the heart muscle) and pericarditis (inflammation of the tissue surrounding the heart) following vaccination.” [5].
Worryingly, NSW Health, Minister Brad Hazzard MP, has urged Year 12 students from high risk LGAs in south-western Sydney to get vaccinated against COVID-19. Mr Hazzard said it was students’ “big chance to really go for gold” [6]. He failed to mention that students will still be able to sit for their HSC even if they choose not to take this 'golden opportunity.'
Professor Ben Marias from The Children's Hospital at Westmead Clinical School and Deputy Director of the Marie Bashir Institute for Infectious Diseases and Biosecurity recently stated, "While unvaccinated students would mingle with their vaccinated peers when school returned, it would not be a major compromising factor." Further, “It’s best for everyone to be vaccinated, but the unvaccinated students would clearly benefit from everyone around them being vaccinated. But the risk to the average 18-year-old child from even the Delta strain is minimal.” [7]
The NSW government itself understands that certain risk factors for COVID-19, irrespective of age, will result in more serious illness and possibly death. Their ‘Enhanced surveillance plan for COVID-19 in NSW’ states that the government must describe “the clinical severity of COVID-19 cases ... as well as characterise risk factors for serious infection to allow for targeted public health strategies.” This is important, they write, because “These indicators identify high-risk groups for severe illness or complications due to SARS-CoV-2 infection to inform targeted public health measures.” [8]
As stated by current Professor, Child & Adolescent Health, Sydney University, Prof. Robert Booy: “This resilience of healthy children begs the question of whether they need to be routinely vaccinated against COVID-19.” [9]
According to TGA reports, ALL of the clinical trials used for their assessments were conducted by vaccine manufacturers and developers and NONE were independent [10], [11], including those informing the recent 'provisional' approval for use on adolescents (conducted by Pfizer) [12]. This same report refers to the use of the Pfizer vaccine on children aged from six months to 12 years, in a clinical trial conducted by BioNTech SE / Pfizer [13].
The government uses modelling by The Peter Doherty Institute to inform their response to COVID-19 [14]. Recently Prof. Jodie McVernon also used their modelling to inform her recommendation to vaccinate adolescents [15].
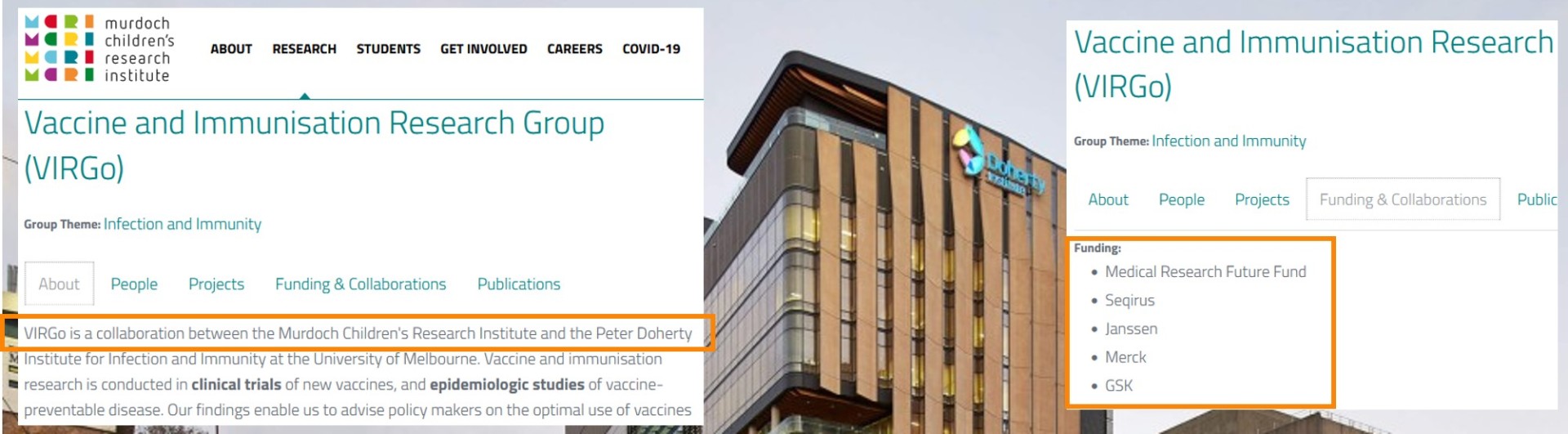
There is a clear conflict of interest here, because The Peter Doherty Institute is also in collaboration with the Vaccine and Immunisation Research Group (VIRGo) / Murdoch Children's Research Institute [15], which has received funding from vaccine manufacturers GSK, Janssen, Merck, Novavax, Sanofi and Sequiris [16].
Prof. Jodie McVernon, spokesperson and researcher for The Peter Doherty Institute for Infection and Immunity, COVID-19 Research [17], also has conflicts of interest with Pfizer and other vaccine manufacturers [18].
The underlying health status of all hospitalised COVID-19 patients in NSW must be clarified if people are to make genuine benefit/risk assessments about the vaccines currently on offer. This is especially vital for young people, for whom the virus presents little threat but vaccination could result in lifelong injury.
Please refer to our recent press release URGENT independent overhaul of government power NEEDED dated August 31st expressing additional concerns.
REFERENCES:
[1] https://www.news.com.au/national/nsw-act/news/covid-warning-for-under-20s-with-several-young-nsw-residents-in-icu/news-story/cfcdc22eefc80f4e2da833968dc0cdf1 : https://amp.smh.com.au/politics/federal/vaccinate-the-young-modelling-backs-strategy-to-slow-virus-spread-20210803-p58fg3.html
[3] COVID-19 Australia: Epidemiology Report 33: Fortnightly reporting period ending 17 January 2021: https://www1.health.gov.au/internet/main/publishing.nsf/Content/C50CAE02452A48A7CA2587320081F7BF/$File/covid_19_australia_epidemiology_report_33_fortnightly_reporting_period_ending_17_january_2021.pdf
[5] FDA announces increase risk of myocarditis (inflammation of the heart muscle) and pericarditis (inflammation of the tissue surrounding the heart) following vaccination from Pfizer vaccine:
[10] AstraZeneca / Oxford: ChAdOx1-S Feb 2021 (9 clinical trials) https://www.tga.gov.au/auspar/auspar-chadox1-s
- 5 were conducted by the vaccine’s developer; the University of Oxford
- 3 were conducted by AstraZeneca
- 1 was conducted by vaccine manufacturer Serum Institute of India/Indian Council of Medical Research
- 3 were conducted by AstraZeneca
- 1 was conducted by vaccine manufacturer Serum Institute of India/Indian Council of Medical Research
[11] Pfizer / BioNTech: Comirnaty BNT162b2 (mRNA) Jan 2021 (2 clinical trials) https://www.tga.gov.au/auspar/auspar-bnt162b2-mrna-comirnaty
- 2 were conducted by vaccine developer BioNTech
[12] Pfizer / BioNTech: Comirnaty BNT162b2 (mRNA) Jul 2021 https://www.tga.gov.au/sites/default/files/auspar-bnt162b2-mrna-210722.pdf
The sponsor also submitted the six month safety data following the second dose as part of the post approval commitment to the original provisional approval for individuals older than 16 years of age.
[13] Clinical Trial No. C4591001, referred to in AUSPAR report, uses 6 months to 12 years of age, provided by Pfizer vaccine developer and manufacturer, due for completion September 23, 2023 https://clinicaltrials.gov/ct2/show/NCT04816643
[14] Doherty Institute: https://www.doherty.edu.au/news-events/news/covid-19-modelling-papers
[15] Prof. Jodie McVernon's recent announcement: https://www.doherty.edu.au/news-events/news/doherty-institute-modelling-report-for-national-cabinet
[16] Murdoch Children’s Research Institute funding & collaboration through VIRGo: https://www.mcri.edu.au/research/themes/infection-and-immunity/vaccine-and-immunisation-research-group-virgo Click on "Funding & Collaborations" for funding; and https://imoparty.com/COI-References Ref 71, for screenshots.
[18] Jodie McVernon – detailed COI can be found at https://imoparty.com/Press-Release-COVID-19-vaccines-set-for-Australia
- Director of Influenza Specialist Group https://www.health.gov.au/sites/default/files/atagi-conflict-of-interest-disclosures.pdf, which received funding from vaccine manufacturers Abbott, GSK, Seqirus, Pfizer, Roche and Sanofi http://www.isg.org.au/index.php/about/sponsorship-and-support/
- Investigator on clinical trials funded by vaccine manufacturers GlaxoSmithKline, bioCSL, Novartis and Pfizer https://www.health.gov.au/sites/default/files/atagi-conflict-of-interest-disclosures.pdf.
- Member of Asia-Pacific Alliance for the Control of Influenza (APACI) https://www.apaci.asia/about-apaci/membership, which has received funds from vaccine manufacturers IFPMA, Roche and Seqirus/CSL https://www.apaci.asia/about-apaci/support-grants
- Member of ATAGI's COVID-19 Working Group https://www.health.gov.au/committees-and-groups/australian-technical-advisory-group-on-immunisation-atagi-covid-19-working-group#atagi-covid19-working-group-members, which provides advice to the Minister for Health on the immunisation program for COVID-19 vaccines as they become available in Australia.
- Investigator on clinical trials funded by vaccine manufacturers GlaxoSmithKline, bioCSL, Novartis and Pfizer https://www.health.gov.au/sites/default/files/atagi-conflict-of-interest-disclosures.pdf.
- Member of Asia-Pacific Alliance for the Control of Influenza (APACI) https://www.apaci.asia/about-apaci/membership, which has received funds from vaccine manufacturers IFPMA, Roche and Seqirus/CSL https://www.apaci.asia/about-apaci/support-grants
- Member of ATAGI's COVID-19 Working Group https://www.health.gov.au/committees-and-groups/australian-technical-advisory-group-on-immunisation-atagi-covid-19-working-group#atagi-covid19-working-group-members, which provides advice to the Minister for Health on the immunisation program for COVID-19 vaccines as they become available in Australia.
